Maipl Therapeutics Reports Preclinical Progress for MA-4604, a Novel Oral Non-Hormonal Treatment of Endometriosis
PR Newswire
SCARSDALE, N.Y., May 14, 2026
SCARSDALE, N.Y., May 14, 2026 /PRNewswire/ — Maipl Therapeutics today highlighted continued preclinical progress for MA-4604, its lead investigational therapy for endometriosis. MA-4604 is a novel oral non-hormonal prostaglandin F2α receptor (FP) antagonist being developed to address the significant unmet medical needs of women living with endometriosis.
The program was recently highlighted in a Drug Hunter review* following the inaugural Health Executive & Research Summit (HERS), where emerging therapeutic approaches for women’s health were showcased.
Endometriosis affects an estimated 10% of women of reproductive age worldwide and is commonly associated with severe pelvic pain. Current treatment options are primarily limited to hormonal therapies that suppress estrogen production or broad anti-inflammatory agents such as NSAIDs, both of which may be associated with effectiveness and tolerability limitations and long-term treatment challenges.
A Targeted, Non-Hormonal Approach
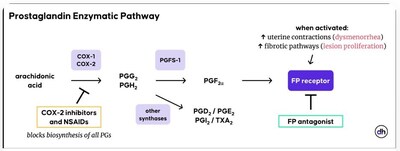
MA-4604 is designed to selectively antagonize the prostaglandin F2α receptor (FP), offering a differentiated mechanism intended to address key pathological drivers of endometriosis without altering hormone levels.
Prostaglandin F2α is a well-characterized mediator of uterine contractions, menstrual pain, and fibrotic signaling associated with endometriosis. By selectively blocking the FP receptor downstream in the prostaglandin pathway, MA-4604 may reduce dysmenorrhea and lesion-associated pathology while potentially avoiding the broader prostaglandin suppression and side effects associated with non-selective NSAIDs.
Therapeutic Hypothesis of FP Antagonism in Endometriosis: Activation of the FP receptor is associated with increased uterine contractility and activation of fibrotic pathways that may contribute to lesion proliferation. FP antagonists are designed to selectively inhibit this signaling pathway to reduce symptoms and disease progression.
Preclinical Results and Development Pathway
Maipl’s MA-4604, which is in-licensed from Ferring Pharmaceuticals, has demonstrated encouraging preclinical activity across multiple translational models:
- Uterine Contractility: Demonstrated efficacy in a rat intrauterine pressure model.
- Pain Reduction: Showed significant activity in F2α-induced acute pain models.
- Disease Modification: Demonstrated efficacy in a mouse model of endometriosis, supporting potential disease-modifying activity.
MA-4604 is currently advancing through IND-enabling studies, with Maipl Therapeutics targeting initiation of first-in-human clinical studies in 2027.
About Maipl Therapeutics
Maipl Therapeutics is a biotechnology company focused on repurposing established targets and developing novel therapies for chronic inflammatory conditions. By leveraging deep biological insights into fibrotic and inflammatory signaling, the company aims to deliver transformative, non-hormonal treatments for women’s health. For more information, visit www.maipltx.com/
Forward Looking Statements
This press release contains forward looking statements, including but not limited to statements regarding the anticipated benefits of the alliance, planned clinical studies and the potential of MA 4604. These statements are subject to risks and uncertainties that could cause actual results to differ materially. Maipl undertakes no obligation to update any forward looking statements.
¹ * https://marketing.drughunter.com/thank-you/endometriosis-inspires-re-examination-of-known-targets
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/maipl-therapeutics-reports-preclinical-progress-for-ma-4604-a-novel-oral-non-hormonal-treatment-of-endometriosis-302771714.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/maipl-therapeutics-reports-preclinical-progress-for-ma-4604-a-novel-oral-non-hormonal-treatment-of-endometriosis-302771714.html
SOURCE Maipl Therapeutics, Inc.